C. Dahmann: Interplay between mechanical tension and cytoskeletal organization in cell separation at compartment boundaries in Drosophila

Principal investigator
Prof. Dr. Christian Dahmann
Technische Universität Dresden
Institute of Genetics
01062 Dresden
Tel: +49 (0)351-463-39537 (office)
christian.dahmann(at)tu-dresden.de
SPP funded collaborator
Jing Wang
Technische Universität Dresden
Institute of Genetics
01062 Dresden
jing.wang4(at)mailbox.tu-dresden.de
Summary
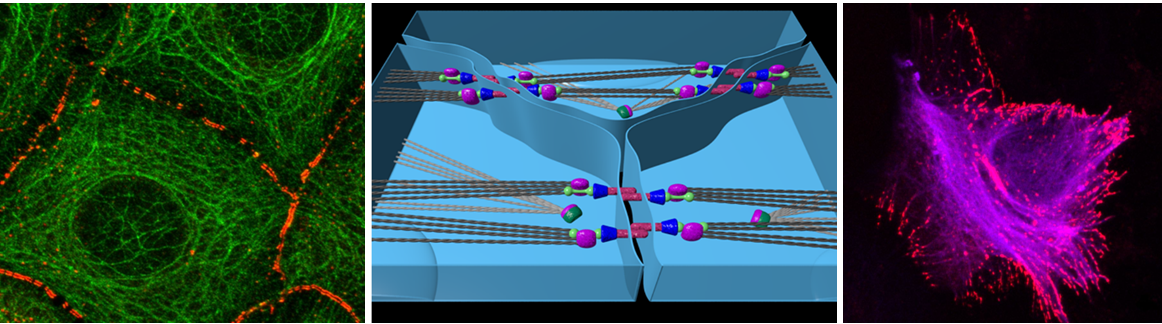
The interplay between mechanical tension and cytoskeletal organization is important for tissue development and homeostasis. Mechanical forces acting on cell junctions can, for example, result in the assembly of cortical F-actin and the recruitment of Myosin motor proteins to the cell cortex. A hallmark of many developing tissues is the subdivision into immiscible groups of cells with distinct functions and fates termed compartments. Compartmentalization is important for the stable positioning of signaling centers within tissues and thus for growth and patterning. The separation of cells from neighboring compartments at boundaries is challenged by cell intercalations caused by cell proliferation and tissue morphogenesis. Recent data show a key role for a local increase in mechanical tension at cell junctions along compartment boundaries in keeping cells from neighboring compartments separated. Local increases in mechanical tension bias cell rearrangements during cell intercalations to maintain straight boundaries and to prevent mixing between cells from neighboring compartments.
Mechanical tension at cell junctions results from actomyosin-based contractile forces and cell adhesion. Local increases in mechanical tension at compartment boundaries correlate with increases in F-actin and non-muscle Myosin II at cell junctions. It has been proposed, but not tested, that F-actin and Myosin II form a multi-cellular cable along compartment boundaries instrumental for separating cells from neighboring compartments. Moreover, whether mechanical tension influences cytoskeletal organization of cells at compartment boundaries and whether such a cytoskeletal reorganization feeds back on mechanical tension has not been addressed. Here, using the Drosophila abdominal epidermis as a model system, we will address these questions. We expect that our results will provide novel insights into the interplay between mechanical forces and biochemical signals involved in organizing cells into functional tissues.
Expertise
Our lab combines genetic, cell biological, and biophysical approaches to understand how cells form functional epithelial tissues. In particular, we use live imaging followed by quantitative image analysis to investigate cellular dynamics in wild-type and mutant wing imaginal discs and the abdominal epidermis of Drosophila. Moreover, laser ablation of cell junctions is used to measure mechanical forces in epithelia.