R. Leube: Desmosomen-Zytoskelett Dynamik

Principal investigator
Prof. Dr. Rudolf Leube
University Hospital Aachen
MOCA
Pauwelsstraße 30
52074 Aachen
Tel: +49 (0)241-8089107 (office)
rleube(at)ukaachen.de

SPP funded collaborator
Dr. Marcin Moch
University Hospital Aachen
MOCA
Pauwelsstraße 30
52074 Aachen
Summary
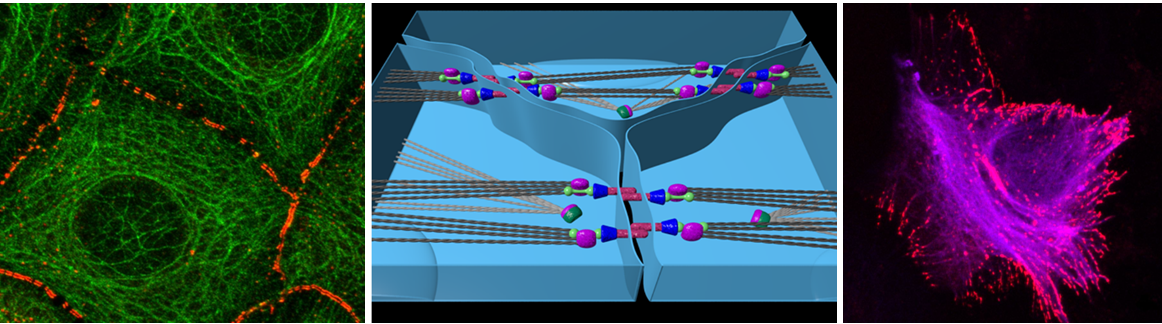
The cytoplasmic keratin intermediate filaments together with their desmosomal anchorage sites at cell-cell borders are hallmark features of epithelial differentiation. The keratin cytoskeleton thereby establishes a transcellular scaffold that affects all aspects of epithelial function requiring a highly dynamic regulation of keratin-desmosome interactions. The central function of the keratin-desmosome complex is evidenced by many genetic and acquired epithelial diseases, in which either keratin polypeptides or desmosomal components are modified. Although molecular binding sites between desmosomal components and keratin polypeptides have been identified, the consequences of desmosomal anchorage for motility, turnover and mechanical properties of keratin filaments have not been studied in detail. Even less is known how the different states of desmosomal adhesion, which have been classified as calcium-sensitive and calcium-insensitive (hyperadhesive), and how desmosomal signaling affects the keratin cytoskeleton. Instead, most of our current knowledge on keratin filament dynamics is based on observations in isolated single cells, which do not reflect the native tissue context. The proposed work therefore aims to fill the existing gap by comparing keratin filaments that are anchored to desmosomes of different adhesive strength with those that are not.
To do this, we will examine the keratin cytoskeleton in human immortalized HaCaT keratinocyte-derived cell lines with no desmosomes, calcium-sensitive desmosomes, hyperadhesive desmosomes, ectopic desmosomal plaques and modified desmosomal signaling. Using advanced microscopy techniques, image analysis routines and tools for measurements of cell mechanics that we have developed for single epithelial cells we want to examine how desmosomal anchorage affects keratin filament branching and bundling, keratin filament motility, keratin filament turnover and cytoplasmic viscoelasticity. We also want to examine how desmosome-mediated mechanical stress affects keratin network morphology and dynamics.
Expertise
Research at MOCA concentrates on the intermediate filament cytosekeleton and its interaction with junctional complexes using genetic and biochemical tools to manipulate cells (2D and 3D cell culture) and organisms (mouse and C. elegans). We employ microscopy to analyze and monitor these interactions in various constellations at different temporal (imaging of fixed specimen and time-lapse recording of vital specimen) and spatial resolution (epifluorescene, confocal and light sheet microscopy and electron microscopy).