M. Matis: Mechanobiological Analysis of Microtubule-Based Forces at Epithelial Adherens Junctions During Tissue Morphogenesis

Principal investigator
Dr. Maja Matis
University of Münster
Institute of Cell Biology
Developmental Mechanobiology Group
Von-Esmarch-Straße 56
48149 Münster
Tel: +49 (0)251-83-57183 (office)
matism(at)uni-muenster.de

SPP funded collaborator
Amrita Singh
University of Münster
Institute of Cell Biology
Developmental Mechanobiology Group
Von-Esmarch-Straße 56
48149 Münster
Tel: +49 (0)251-83-52187 (office)
amrita.singh(at)uni-muenster.de
Summary
During tissue morphogenesis, individual cells self-assemble into complex tissues and organs with highly specialized forms and functions. Such precise sculpting requires the application of forces generated within cells by the cytoskeleton and transmission of these forces through adhesion molecules within and between neighboring cells.
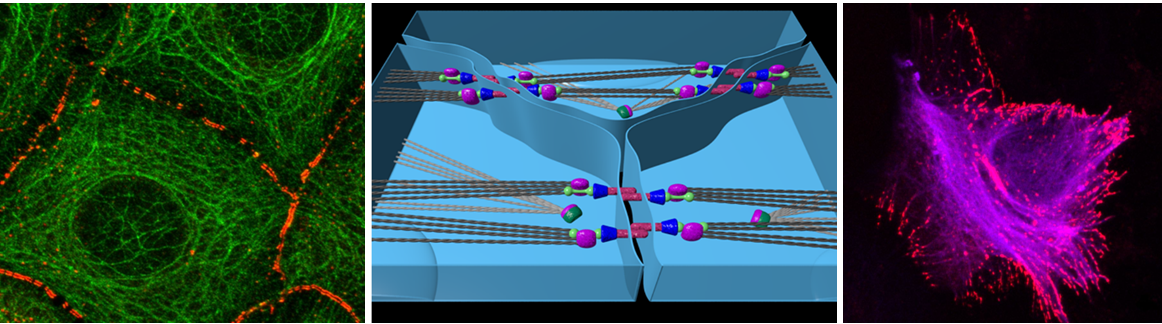
In order to reshape a tissue, force generation must exceed mechanical resistance, thus global patterns of force generation and tissue stiffness jointly dictate speed and direction of tissue rearrangements. While many of the genes and chemical cues that regulate these processes during morphogenesis have been identified, little is known quantitatively about how these mechanical processes are coupled across cells in a developing tissue. Despite growing evidence that MT can generate forces in cells in a manner analogous to actin, relatively little is known about how coupling of MT-based forces at epithelial intercellular junctions contributes to the mechanical state of tissue and cell-shape changes during morphogenesis.
The goal of this project is to develop and apply new tools that will help answering the fundamental question how cells coordinate forces across a tissue during morphogenetic rearrangements. We will use a dual approach that relies on new optical and chemical tools that, in conjunction with classical genetic approaches, will be used to quantitatively analyze how the cytoskeleton coordinates forces across cells during morphogenetic tissue rearrangements. Together, this interdisciplinary approach will allow us to quantitatively address the fundamental principles underlying force-control during tissue organization via MTs. We expect that insights from our study will uncover new roles of the MT cytoskeleton in epithelial tissue morphogenesis, and open up new inroads to investigate how derangement of MT dynamics at intercellular junctions leads to diseases.
Expertise
Our lab investigates the mechanical and structural properties of apical non-centrosomal MTs nucleated at adherens junctions. For this we are using new optical and chemical tools (e.g live imaging, caged MT drugs, laser ablation, FRAP, FRET based tension sensors, etc.) in conjunction with classical genetic and proteomic approaches. The quantitative data obtained in this interdisciplinary approach are correlated with results obtained from quantitative image analysis of developing epithelium to study how mechanobiological properties of MTs contribute to tissue remodeling.