P. Strnad: Consequences of desmosomal alterations in the intestinal epithelia

Principal investigator
PD Dr. Pavel Strnad
University Hospital Aachen
Department of Internal Medicine III
Pauwelsstraße 30
52074 Aachen
Tel: +49 (0)241-80-35324 (office)
pstrnad(at)ukaachen.de

Associated collaborator
Assoc. Prof. Dr. Peter Boor
University Hospital Aachen
Institute of Pathology
Pauwelsstraße 30
52074 Aachen
Tel: +49 (0)241-80-85227 (office)
pboor(at)ukaachen.de

Associated collaborator
Dr. Sonja Djudjaj
University Hospital Aachen
Institute of Pathology
Pauwelsstraße 30
52074 Aachen
sdjudjaj(at)ukaachen.de

SPP funded collaborator
Annika Gross
University Hospital Aachen
Department of Internal Medicine III
Pauwelsstraße 30
52074 Aachen
angross(at)ukaachen.de
Summary
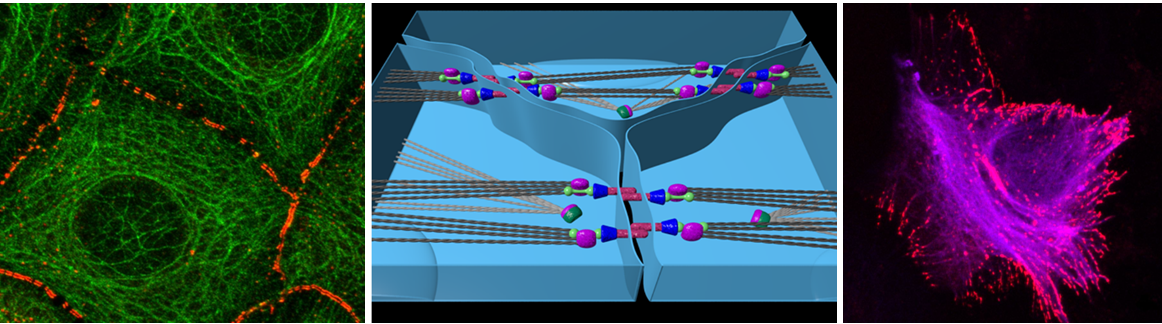
To protect the organism against luminal pathogens while enabling selective uptake of nutrients through specific transport systems, intestinal epithelia contain the apical junctional complex (AJC) consisting of tight junctions, adherens junctions and desmosomes. Desmosomes constitute the least investigated AJC component and are composed of transmembrane cadherins of the desmoglein (Dsg) and desmocollin (Dsc) type that are associated through the intracellular plaque proteins plakoglobin, plakophilin and desmoplakin to the cytoplasmic keratin intermediate filament cytoskeleton. Dsg2 and Dsc2 are the major desmosomal cadherins of intestinal epithelia. They have been implicated in the regulation of epithelial cell proliferation and tumorigenesis. Recent data indicate that Dsg2 is reduced in patients with inflammatory bowel disorders and that stabilization of inflammation-induced Dsg2 loss may ameliorate a loss of epithelial barrier function. To further study the functional relevance of Dsg2 in the intestinal epithelia, we generated intestine-specific, conditional DSG2 knockouts (DSG2 Δint/Δint) Gavage with FITC-dextran and subsequent FITC measurement in serum was used to assess intestinal permeability. Colitis was induced by five-day treatment with dextran sodium sulfate (DSS) or gavage-mediated infection with Citrobacter rodentium. DSG2 Δint/Δint mice display a robust knockdown of intestinal Dsg2 and a profound alteration of the remaining desmosomal components with upregulation of Dsc2 and a decrease in plakoglobin and desmoplakin while overall histology is inconspicuous. Under basal condition, DSG2 Δint/Δint mice are phenotypically normal but have a somewhat increased intestinal permeability. After DSS treatment, they suffer higher weight loss, and stronger epithelial damage/inflammation. 14 days after infection with Citrobacter rodentium, DSG2 Δint/Δint mice display stronger crypt hyperplasia and elevated levels of the proinflammatory/bacterial response genes. In our current proposal, we are assessing the pathomechanisms underlying the observed changes using genetically modified mice and intestinal organoids as the primary research tools.
Expertise
The group of PD Dr. Strnad studies the development and progression of chronic digestive diseases using transgenic animals and primary cell cultures as the major research models. The typical methods that are routinely used include a large variety of experimental murine digestive disease models, thorough histological/immunohistochemical analysis, microarray/omics approaches and basic biochemical methods (immunoprecipitation, subcellular fractionation, analysis of oxidative stress etc). Human relevance of the observed experimental findings is further evaluated in patients with digestive diseases that are either recruited in University Hospital Aachen or are available through a network of collaborating scientists.